
Welcome to the Laboratory of Molecular Chronobiology! We study the molecular bases of circadian rhythms, their connections to physiology, including the immune response, and their implications in various health problems, including schizophrenia and infectious diseases.

Many physiological processes present near-24h oscillations, even in constant conditions (1). For example, the hormone cortisol is at high levels in the blood in the morning, and low levels in the evening; another well-defined rhythm is that of body temperature, which in human is high in the day and at its lowest point in the late night. These daily rhythms are called circadian rhythms, and they are generated by circadian clocks. The research in the Laboratory of Molecular Chronobiology aims at deciphering how these clocks work, and more specifically, to find an answer to three questions: What are the mechanisms or gears of these biological clocks? How can these clocks control physiology? How are these clocks controlled by the environment? What happens when the clocks are broken?
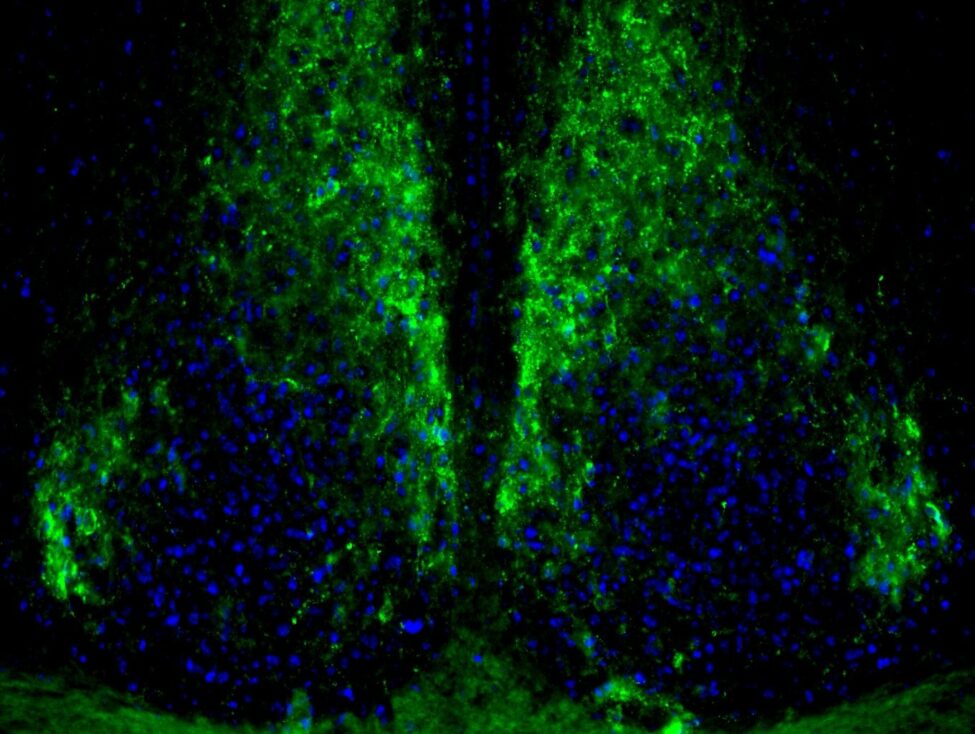
The master circadian clock in mammals such as mice or humans is located in a brain region called the suprachiasmatic nucleus (figure). However, recent research has shown that clocks are also present in other brain regions and in most peripheral organs too. During evolution, circadian clocks have evolved by giving an advantage to living organisms, by adapting their physiology to the cyclically changing environment (e.g. light/dark, warm/cold daily cycles), and by organizing in time the processes occurring in various tissues and within cells. Thus, it is not a surprise that disturbances of circadian clocks have been associated with various diseases, including cancer, metabolic diseases and mood disorders.
1. The identification and study of clock components
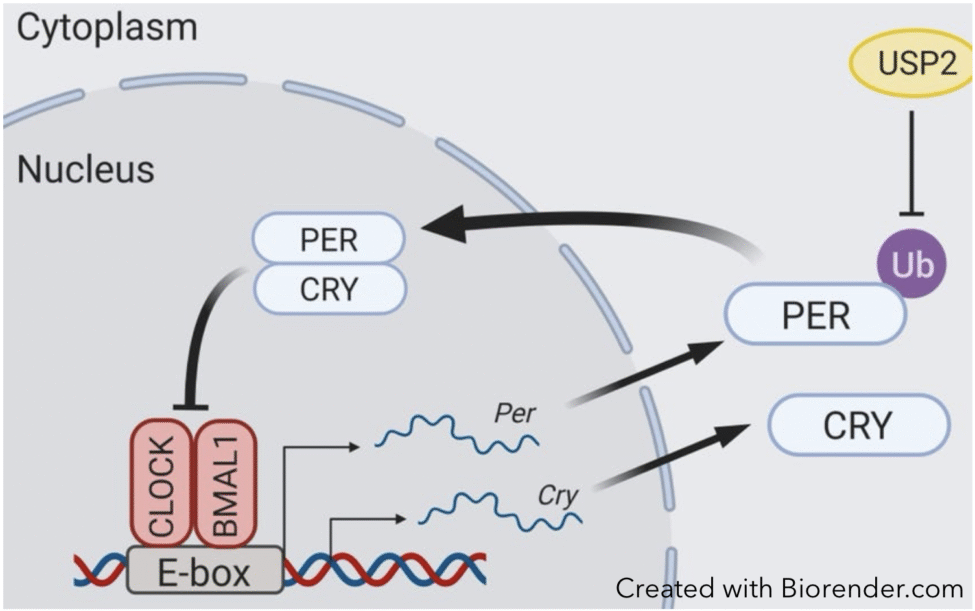
Circadian clocks are made up of so-called clock genes (1), and animals or human subjects with mutated clock genes present rhythm disturbances, even arrhythmicity in some cases. Over the years, the work in the laboratory has led to the identification of new clock genes. One example of this is the finding that proteins of the REV-ERB and ROR families can act as clock components (2, 3). More recently, we have been interested in the circadian role of USP2, a protein known to remove ubiquitin tags from proteins. Indeed, tagging of clock proteins with ubiquitin, and the subsequent degradation of these tagged proteins, has been shown to be critical for circadian timing (4). We now found that the reverse reaction (removal of the ubiquitin tag) is important for the clock too: USP2 binds to and deubiquitinates the clock protein PER1, and mice devoid of USP2 present abnormal rhythms (5). USP is also involved in the response of the clock to light (6).
2. The circadian control of the immune response and infectious diseases
Circadian clocks are found everywhere in the body, including in cells of the immune system (7). Does it mean that immunity is more or less effective according to time of day? In collaboration with Dr. Nathalie Labrecque (IRCM, Université de Montréal), we found that this was the case: the response to vaccination varies according to time of day. When mice receive this treatment in the middle of the day, their T cells respond more strongly to antigen presentation than at any other times of day or night (8, 9). Mice lacking a clock within their T cells do not exhibit this circadian rhythm. Our analysis of molecular rhythms in T cells has then allowed us to conclude that these cells are preset (by their own intrinsic clock) to be ready to be activated efficiently in the daytime, and to be more resistant to vaccination at night (9, 10).
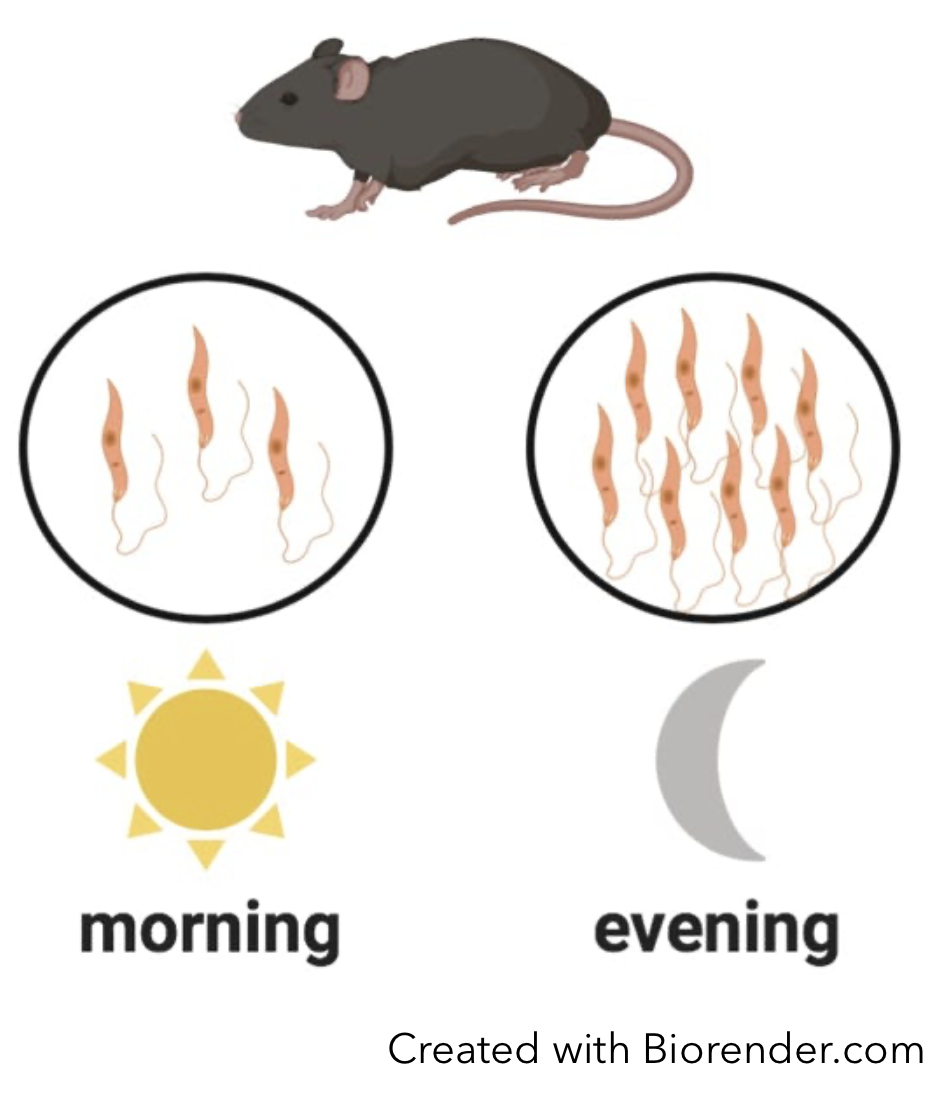
What is the impact of rhythms in the immune system for the fight against infectious diseases? In collaboration with Dr. Martin Olivier (RI-MUHC, McGill University), we aim to answer this question in the context of parasitic infections (11, 12). The parasite Leishmania causes a serious disease prevalent particularly in tropical regions, leishmaniasis. We discovered that the immune response varies according to the time of day at which the parasite is inoculated in mice (figure): infection in the evening leads to a stronger mobilization of immune cells, and since the parasite requires these cells for its development, an evening infection is beneficial for the parasite, which is replicating more efficiently (13). We have also shown a circadian rhythm of the infection by Plasmodium, the parasite causing malaria (14, 15). For both parasites, the clocks of immune cells, in particular macrophages, is important for the circadian control (10, 15, 16).
3. Circadian disruption and schizophrenia
Up to 80% of patients with schizophrenia show disrupted sleep and circadian rhythms (17, 18). In collaboration with Dr. Lalit Srivastava (Douglas Research Centre, McGill University), we are addressing two questions. First, what is the cause of this circadian rhythm disruption? Second, could these disrupted rhythms worsen schizophrenia symptoms, or act as a risk factor for the disease, and other disorders due to altered brain development (17, 18)? To address these questions, we use mouse models. For example, we showed that mice with a mutation in the Dysbindin-1 gene (which was associated with schizophrenia) present abnormal circadian rhythms (19). Moreover, lighting conditions that disrupt the mouse circadian rhythms exacerbate certain schizophrenia-related behaviours in the mutant mice (19, 20). We have also studied a mouse model of prenatal infection, which is a known risk factor for brain development disorders: in this case too, altered circadian rhythms are seen, and circadian disruption are associated with features of the diseases (21, 22). This work might allow developing novel strategies, taking circadian rhythms into consideration, for improving cognition in schizophrenia and other psychiatric conditions. Actually, we are now using the mouse data to investigate the risk for psychiatric disorders in human populations (with Dr. Patricia Silveira, Douglas, McGill).

4. The regulation of human circadian clocks: implications for shift work
In order to study the regulation of human circadian clocks, we teamed up with Dr. Diane Boivin (Douglas Research Centre, McGill University). By simulating night shift work in the laboratory, and in field studies on actual shift workers, we showed that similarly to the central clock, human peripheral clocks do not adapt well to a night shift (23, 24). The consequence is a loss of synchronization of the molecular (23-25), immune (26) and metabolic (27, 28) rhythms. Furthermore, we made the first demonstration of a pharmacological resetting of peripheral clocks in humans (29). Finally, we also looked at clock gene expression in post-mortem brain tissue, and found that rhythms of clock gene RNAs differed between the brains of Alzheimer’s disease patients compared to those of control subjects, which might underlie the sleep and circadian rhythms of these patients (30).
1. Duguay, D. & Cermakian, N. (2009) Chronobiol Int 26, 1479-513. 2. Guillaumond, F., Dardente, H., Giguère, V. & Cermakian, N. (2005) J Biol Rhythms 20, 391-403. 3. Mongrain, V., Ruan, X., Dardente, H., Fortier, E. E. & Cermakian, N. (2008) Genes Cells 13, 1197-210. 4. Stojkovic, K., Wing, S.S., Cermakian, N. (2014) Front Mol Neurosci, 7:69. 5. Yang, Y. et al. (2012) Biol Open, 1:789-801. 6. Srikanta, S.B., (…) Cermakian, N. (2025) J Neurochem, 169:e70018. 7. Nobis, C.C., Labrecque, N., Cermakian, N. (2018) Curr Opin Physiol, 5:90-98. 8. Fortier, E.E. et al. (2011) J Immunol 187:6291-300. 9. Nobis, C.C., (…) Cermakian, N. (2019) Proc Natl Acad Sci USA, 1;116(40):20077-20086. 10. Cermakian, N., Labrecque, N. (2023) J Immunol, 210:12–18. 11. Carvalho Cabral, P., Olivier, M., Cermakian, N. (2019) The Complex Interplay of Parasites, their Hosts and Circadian Clocks. Front Cell Infect Microbiol, 9:425. 12. Carvalho Cabral, P., Tekade, K., Stegeman, S.K., Olivier, M., Cermakian, N. (2022) Par Immunol, 44:e12903. 13. Kiessling, S., Dubeau-Laramée, G., Ohm, H., Labrecque, N., Olivier, M., Cermakian, N. (2017) Sci Rep, 7:10892. 14. Carvalho Cabral, P., Weinerman, J., Olivier, M., Cermakian, N. (2024) iScience, 27:109684. 15. Carvalho Cabral, P., Richard, V.R., Borchers, C.H., Olivier, M., Cermakian, N. (2024) ImmunoHorizons, 8(6):442–456. 16. Carvalho Cabral, P., Stegeman, S.K., Olivier, M., Cermakian, N. (2024) Par Immunol, 46:e13053. 17. Delorme, T.C., Srivastava, L.K., Cermakian, N. (2020) Journal of Biological Rhythms, 35(4):325-339. 18. Bouteldja, A.A., Penichet, D., Srivastava, L.K., Cermakian, N. (2024) European Journal of Neuroscience, 60:3858–3890. 19. Bhardwaj, S.K., Stojkovic, K., Kiessling, S., Srivastava, L.K., Cermakian, N. (2015) Behav Brain Res, 284:58-68. 20. Cloutier, M.E., Srivastava, L.K., Cermakian, N. (2022) J Biol Rhythms, 37(6):655-672. 21. Delorme, T.C., Srivastava, L.K., Cermakian, N. (2021) Brain Behav Immun, 93:119-131. 22. Delorme, T., (…) Cermakian, N. (2025) Brain Behav Immun, 127:409-422. 23. Cuesta, M., Boudreau, P., Cermakian, N., Boivin, D.B. (2017) Sci Rep, 7:16310. 24. Koshy, A., Cuesta, M., Boudreau, P., Cermakian, N., Boivin, D.B. (2019) FASEB Journal, 33(6):6789-6800. 25. Kervezee, L., Cuesta, M., Cermakian, N., Boivin, D.B. (2018) Proc Natl Acad Sci USA, 115(21):5540-5545. 26. Cuesta, M., Boudreau, P., Dubeau-Laramée, G., Cermakian, N., Boivin, D.B. (2016) J Immunol, 196(6):2466-75. 27. Kervezee, L., Cermakian, N., Boivin, D.B. (2019) PLOS Biol, 18;17(6):e3000303. 28. Kervezee, L., Koshy, A., Cermakian, N., Boivin, D.B. (2023) J Biol Rhythms, 38:64-76. 29. Cuesta, M., Cermakian, N., Boivin, D.B. (2015) FASEB J, 29(4):1360-70. 30. Cermakian, N., Waddington Lamont, E., Boudreau, P. & Boivin, D.B. (2011) J Biol Rhythms 26, 160-170.
This content has been updated on 3 February 2026 at 15h35.